The landscape of the life sciences ecosystem is ever-changing, and laboratory operations continually adapt to meet market demands, with efficiency and productivity crucial to the industry’s evolution. Among the key drivers of this transformation are the adoption of methodologies like the Lean Lab principles and U-shape layout methodologies. These philosophies and strategies stand as vital tools in enhancing operational excellence to address the industry’s increasingly demanding needs.
At Kymos, our commitment to innovation has led us to embrace these methodologies, reshaping our laboratory operations and impacting the delivery of quality services to our clients. In the present article you will find information about the Lean Lab philosophy and the U-shape layout, how Kymos has adopted them, and the upcoming event at our facilities, offering an opportunity to witness these methodologies in action.
Understanding the Lean Lab Philosophy and U-Shape Implementation
The fundamental concept behind Lean philosophy is achieving results efficiently in terms of cost, speed, or both. It aims to reduce resource usage and costs while enhancing productivity and product or service quality. Derived from Lean Manufacturing principles, “Lean Lab” applies this philosophy specifically to laboratory ecosystems.
Implementing the Lean Lab theories involves a fundamental restructuring of laboratory design and workflow optimization. Testing and analytical laboratories often contend with extended and fluctuating lead times due to factors such as high work in process (WIP), poorly designed layouts causing timing issues (e.g., queues in front of each test), and erratic and volatile incoming workloads affecting productivity.
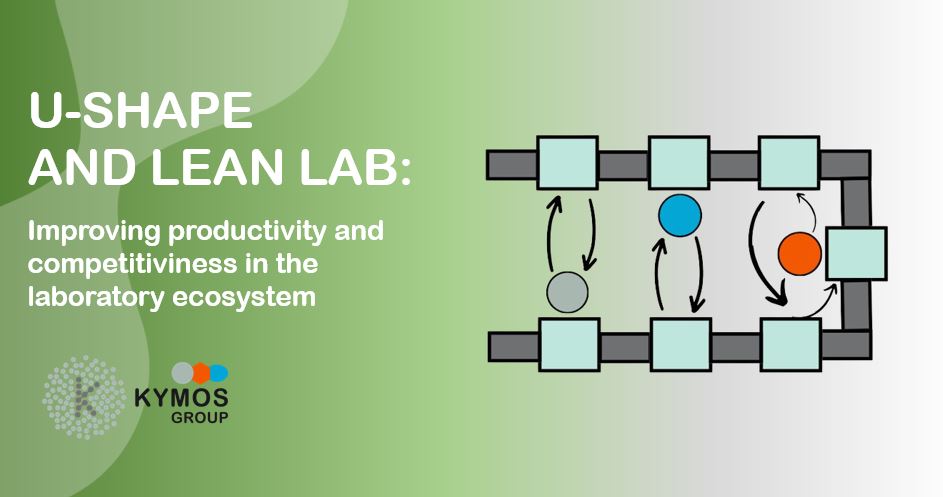
The U-shape layout, a core aspect of Lean Lab philosophy, streamlines workflow by reducing unnecessary movements, minimizing errors, and optimizing space usage. Resembling an assembly line, this layout positions workstations in a U-shaped pattern, promoting smoother processes and improving communication among team members.
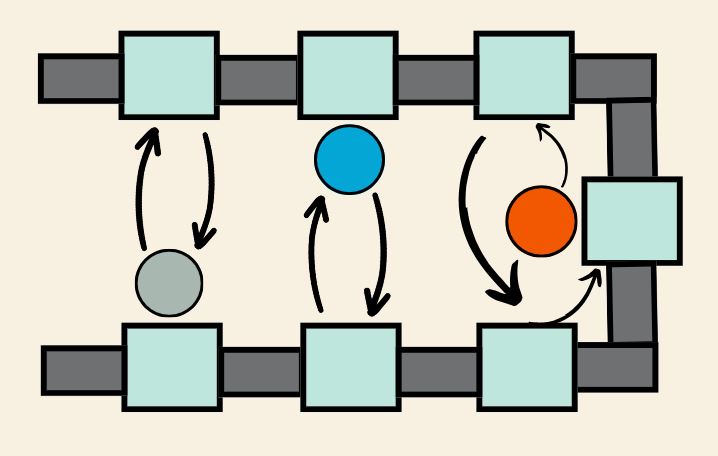
When executed effectively, laboratories can shorten their lead times and decrease their WIP levels through a one-piece flow that creates a straightforward process with consistent workloads. Additionally, the U-shaped cell, equipped with a cross-trained team, augments flexibility, enabling adaptation to demand fluctuations. Team members are prepared and enabled to perform various functions, enhancing responsibility, quality, and traceability.
Enhancing Productivity in Biopharmaceutical Testing at Kymos
Kymos has experienced firsthand the transformative impact of Lean Lab and U-Shape implementation in our biopharmaceutical testing department. Particularly in the batch release of biosimilars for our clients, these methodologies have significantly accelerated our processes while maintaining the highest standards of quality control.
In the projects where the Lean methodology has been applied, we have observed the following results:
- The lead times of experimental work have been reduced up to 70%.
- The productivity of our analysts has multiplied up to x6 (six times more samples per hour).
- The occupied area has been reduced by more than 80% with the new distribution of our lab.
- We have defined our maximum capacity per week (number of batches), thereby determining our exact timings and results.
This optimization has not only enhanced our efficiency but has also empowered us to meet the escalating demand for biosimilar release in the market.
In response to this, Kymos is actively planning to expand our laboratory capacity in the upcoming months. This expansion will improve our capacity, and by applying the principles of Lean Lab and using the U-shape layout we will be able to ensure timely and quality services to our clients and customers.
Upcoming Event the 28/11/2023:
Lean Lab: the Lean system applied to improve laboratory competitiveness
In line with all the previously presented, Kymos together with CataloniaBio & HealthTech and Leanbox is organizing a round table and workshop on the Lean methodology applied to laboratories in the health sector.
This event will focus on practical cases of Lean Lab application in the analytical and testing environment and will count with the presence of key players in the industry.
Attendees will also have the opportunity to visit our facilities at Cerdanyola del Vallès to see firsthand the implementation of this system in a laboratory setting in our biopharmaceutical testing department.
More info and registration: https://www.cataloniabioht.org/events/leanlab-el-sistema-lean-aplicat-a-millorar-la-competitivitat-del-laboratori/?lang=en
Conclusions
The integration of Lean Lab philosophy and the utilization of the U-shape layout in our laboratory has significantly improved our productivity, efficiency, and service quality. We remain committed to leveraging these methodologies to ensure our clients receive the highest standard of service and quality.
If you require assistance with biosimilars release, biopharmaceutical testing or any of your CMC or bioanalytical projects, please do contact us at: https://kymos.com/contact/

