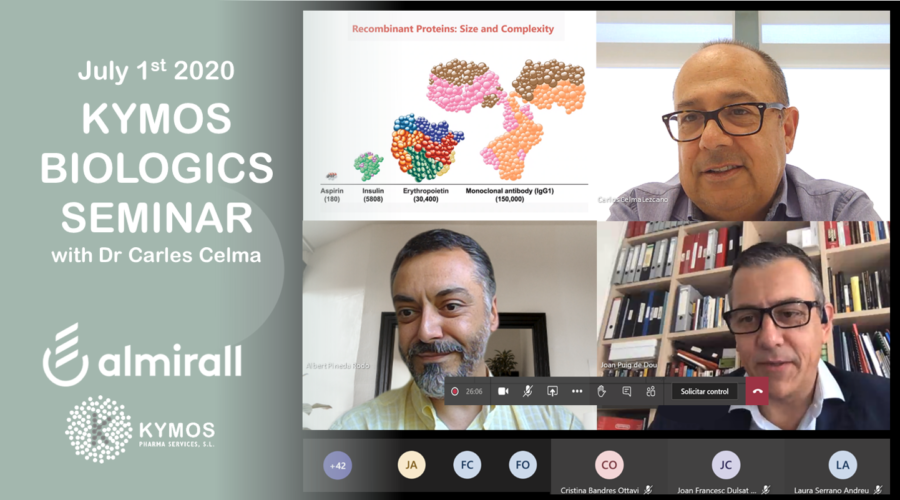
Dr Carles Celma, Scientific Director at KYMOS was invited by ALMIRALL to present a one-off Biologics seminar on July 1st. The initiative, held remotely via Teams due to the COVID-19 pandemic, was promoted by Technical R&D, Almirall R&D, and aimed to explore analytical capabilities for the characterization and quality control of biopharmaceutical products, a subject of growing importance within the healthcare community.
KYMOS is an FDA-inspected European CRO specialized in bioanalysis and CMC of both small and large molecules. Dr Celma is an industry-recognized key opinion leader with decades of experience working with innovative biologics and biosimilars. “The flow of new small-molecule APIs has been slowing down during the last decade while spending on new biological entities is on the rise” states Dr Celma. “With that said, the development of complex biologics such as monoclonal antibodies (mAbs), vaccines or coagulation factors remains analytically challenging, which is why more pharmaceutical companies tend to outsource their characterizations and comparability studies to capable CROs like KYMOS” he concludes.
ALMIRALL is a leading skin-health focused global pharmaceutical company that partners with healthcare professionals, applying science to provide medical solutions to patients and future generations. Their efforts are focused on fighting skin health diseases and helping people feel better. We support healthcare professionals in their continuous improvements, providing our innovative solutions where they are needed.
For more information on ALMIRALL please visit our website or contact us here.
For more information on KYMOS please visit our website or contact us at commercial@kymos.com.